正在加载图片...
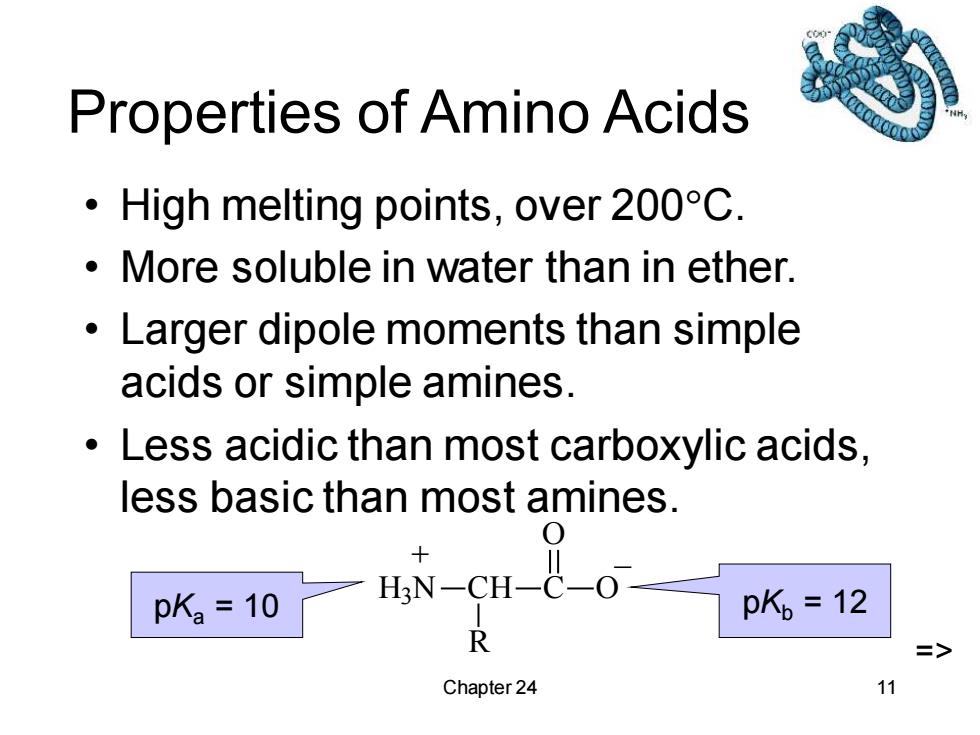
Properties of Amino Acids High melting points,over 200C More soluble in water than in ether. Larger dipole moments than simple acids or simple amines. Less acidic than most carboxylic acids, less basic than most amines. pK2=10 H3N-CH-C-O pK,=12 R 三> Chapter 24 11Chapter 24 11 Properties of Amino Acids • High melting points, over 200C. • More soluble in water than in ether. • Larger dipole moments than simple acids or simple amines. • Less acidic than most carboxylic acids, less basic than most amines. H3 N CH R C O O + _ pKa = 10 pKb = 12 =>