正在加载图片...
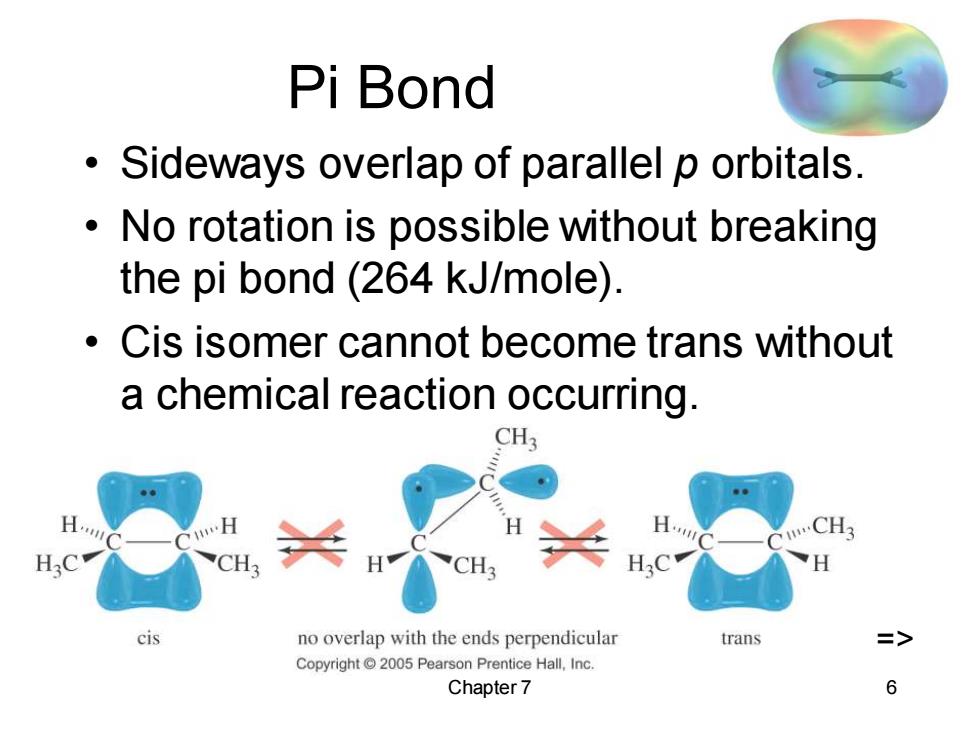
Pi Bond Sideways overlap of parallel p orbitals. No rotation is possible without breaking the pi bond (264 kJ/mole). Cis isomer cannot become trans without a chemical reaction occurring. CH3 A. CH3 H.C H:C H cis no overlap with the ends perpendicular trans Copyright 2005 Pearson Prentice Hall,Inc. Chapter 7 6Chapter 7 6 Pi Bond • Sideways overlap of parallel p orbitals. • No rotation is possible without breaking the pi bond (264 kJ/mole). • Cis isomer cannot become trans without a chemical reaction occurring. =>