正在加载图片...
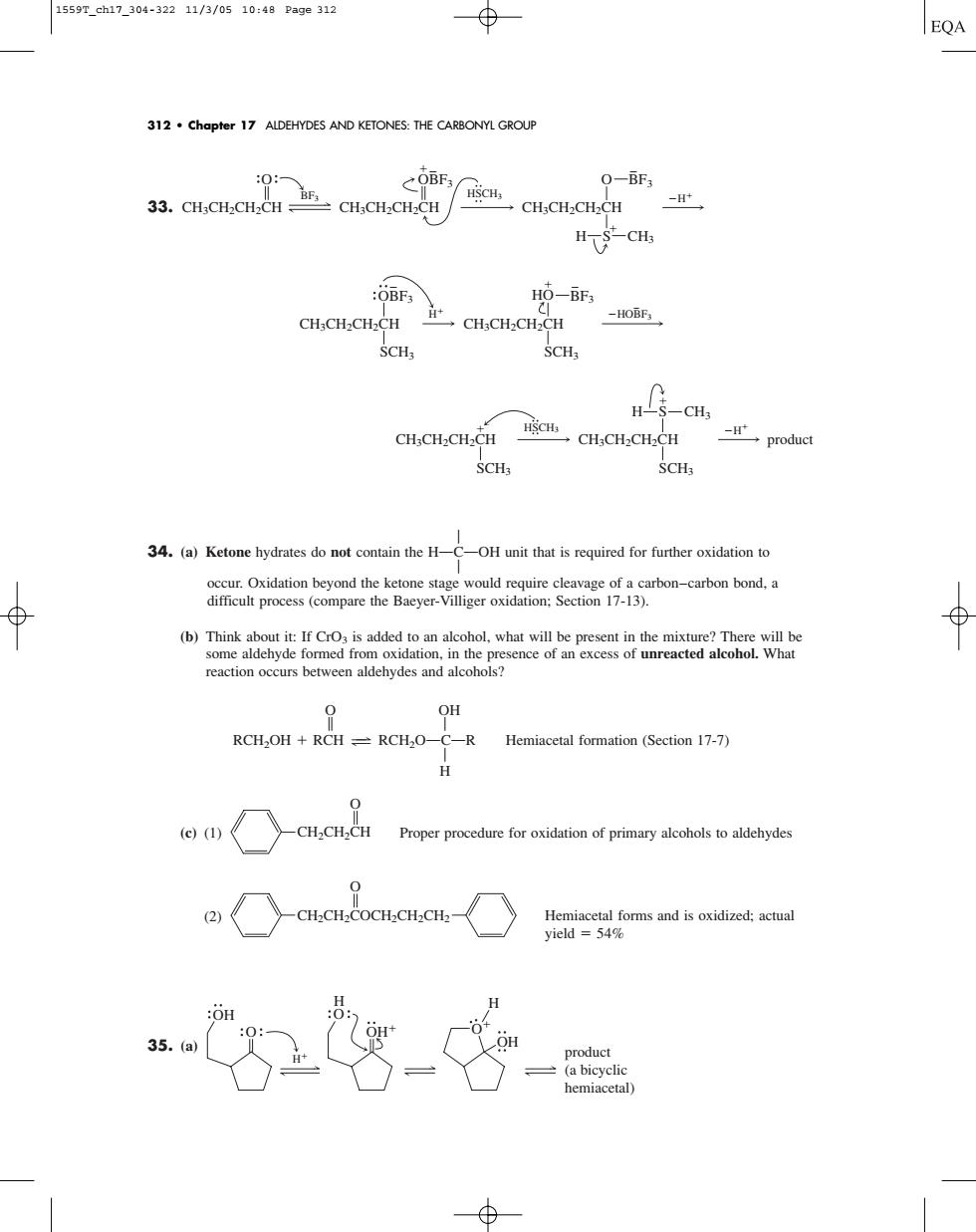
1559T_ch17_304-32211/3/0510:48Page312 EQA 312.Chapter 17 ALDEHYDES AND KETONES:THE CARBONYL GROUF Q-BF3 CH CH-CH-CH HTCH :OBF3 H -HOBF; CH:CH.CH-CH 一CH.CH.CH2CH SCH SCH CH3CH2CH2CH CH.CH-CH.CH product H 34.(a)Ketone hydrates do not contain the H-OH unit that is required for further oxidation to reaction occus between aldehydes and alcohos? 30 RCH OH +RCH-RCH.O C-R Hemiacetal formation (Section 17-7) Proper procedure for oxidation of primary alcohols to aldehydes Hegormsndoiata hemiaceta 33. A 34. (a) Ketone hydrates do not contain the HOCOOH unit that is required for further oxidation to A occur. Oxidation beyond the ketone stage would require cleavage of a carbon–carbon bond, a difficult process (compare the Baeyer-Villiger oxidation; Section 17-13). (b) Think about it: If CrO3 is added to an alcohol, what will be present in the mixture? There will be some aldehyde formed from oxidation, in the presence of an excess of unreacted alcohol. What reaction occurs between aldehydes and alcohols? O OH B A RCH2OH RCH 34 RCH2OOCOR Hemiacetal formation (Section 17-7) A H (c) (1) Proper procedure for oxidation of primary alcohols to aldehydes (2) Hemiacetal forms and is oxidized; actual yield 54% 35. (a) H OH O OH H product (a bicyclic hemiacetal) OH O H O CH2CH2COCH2CH2CH2 O CH2CH2CH O SCH3 CH3CH2CH2CH HSCH3 CH3CH2CH2CH SCH3 product H S CH3 H H SCH3 CH3CH2CH2CH OBF3 HOBF3 CH3CH2CH2CH SCH3 HO BF3 BF3 HSCH3 CH3CH2CH2CH CH3CH2CH2CH O OBF3 H CH3CH2CH2CH CH3 O BF3 H S 312 • Chapter 17 ALDEHYDES AND KETONES: THE CARBONYL GROUP 1559T_ch17_304-322 11/3/05 10:48 Page 312���������