正在加载图片...
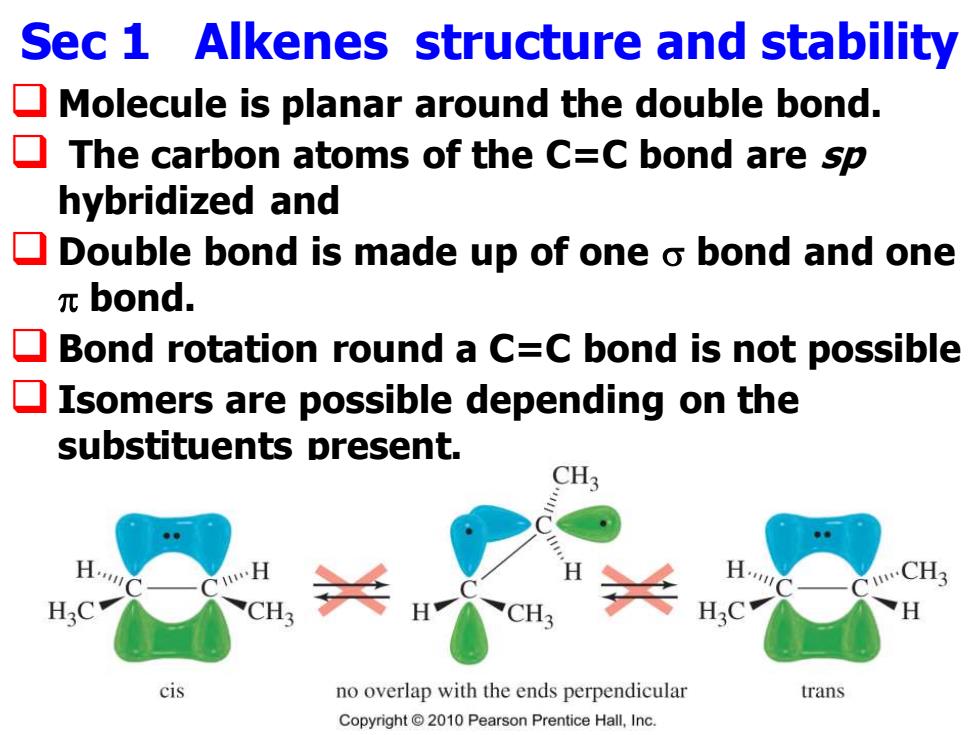
Sec 1 Alkenes structure and stability Molecule is planar around the double bond. The carbon atoms of the C=C bond are sp hybridized and Double bond is made up of one o bond and one πbond. Bond rotation round a C=C bond is not possible Isomers are possible depending on the substituents present. CH3 H. H. CH3 H3C cis no overlap with the ends perpendicular trans Copyright2010 Pearson Prentice Hall,Inc.Sec 1 Alkenes structure and stability ❑ Molecule is planar around the double bond. ❑ The carbon atoms of the C=C bond are sp hybridized and ❑ Double bond is made up of one bond and one bond. ❑ Bond rotation round a C=C bond is not possible ❑ Isomers are possible depending on the substituents present