正在加载图片...
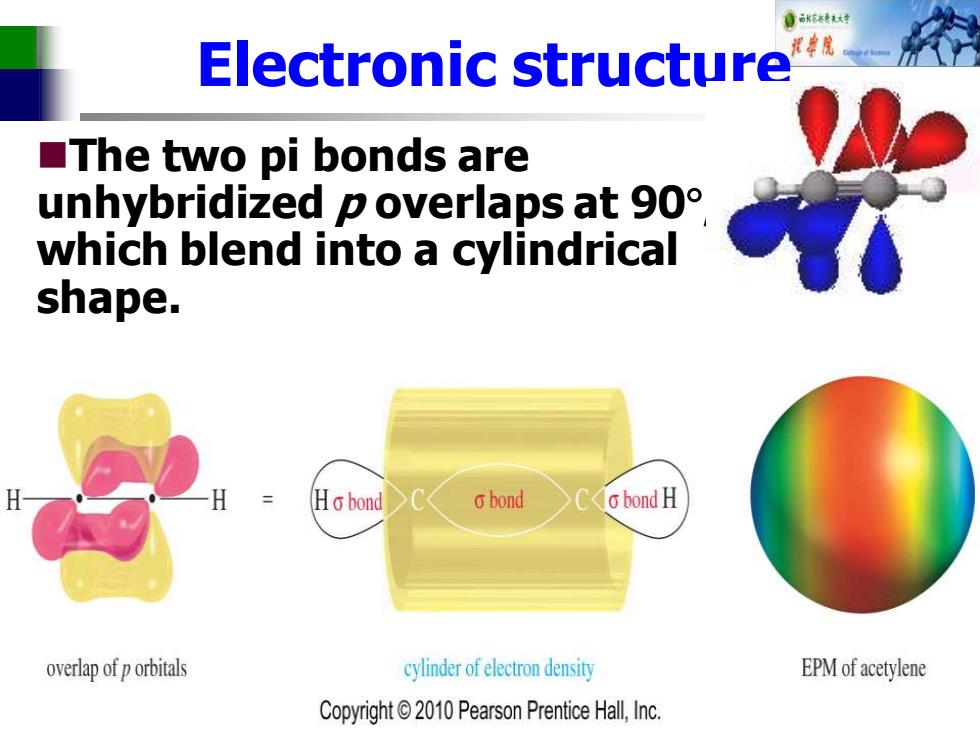
自标特花对 Electronic structure ■The two pi bonds are unhybridized p overlaps at 90 which blend into a cylindrical shape. Ho bond 6bond o bond H overlap of p orbitals cylinder of electron density EPM of acetylene Copyright2010 Pearson Prentice Hall,Inc. Electronic structure ◼The two pi bonds are unhybridized p overlaps at 90 , which blend into a cylindrical shape