正在加载图片...
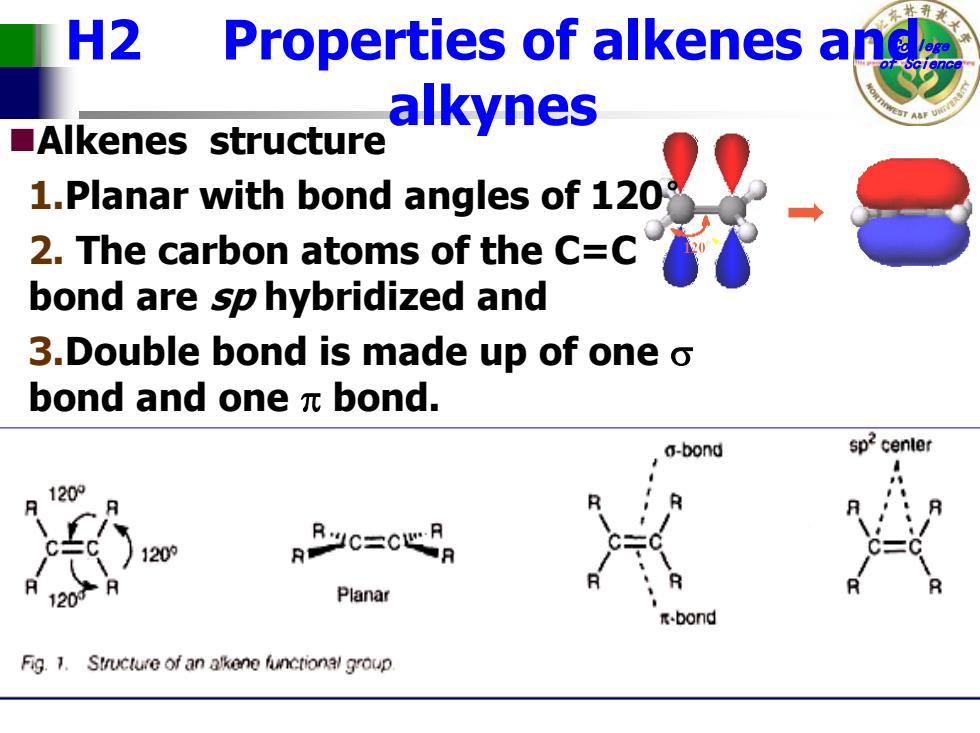
H2 Properties of alkenes and alkynes ■Alkenes structure 1.Planar with bond angles of 120 2.The carbon atoms of the C=C bond are sp hybridized and 3.Double bond is made up of one o bond and one元bond. a-bond 120° 120 120 Planar -bond Fig.1.Structure of an alkene functional groupCollege H2 Properties of alkenes andof Science alkynes Alkenes structure 1.Planar with bond angles of 120 ° 2. The carbon atoms of the C=C bond are sp hybridized and 3.Double bond is made up of one σ bond and one π bond