正在加载图片...
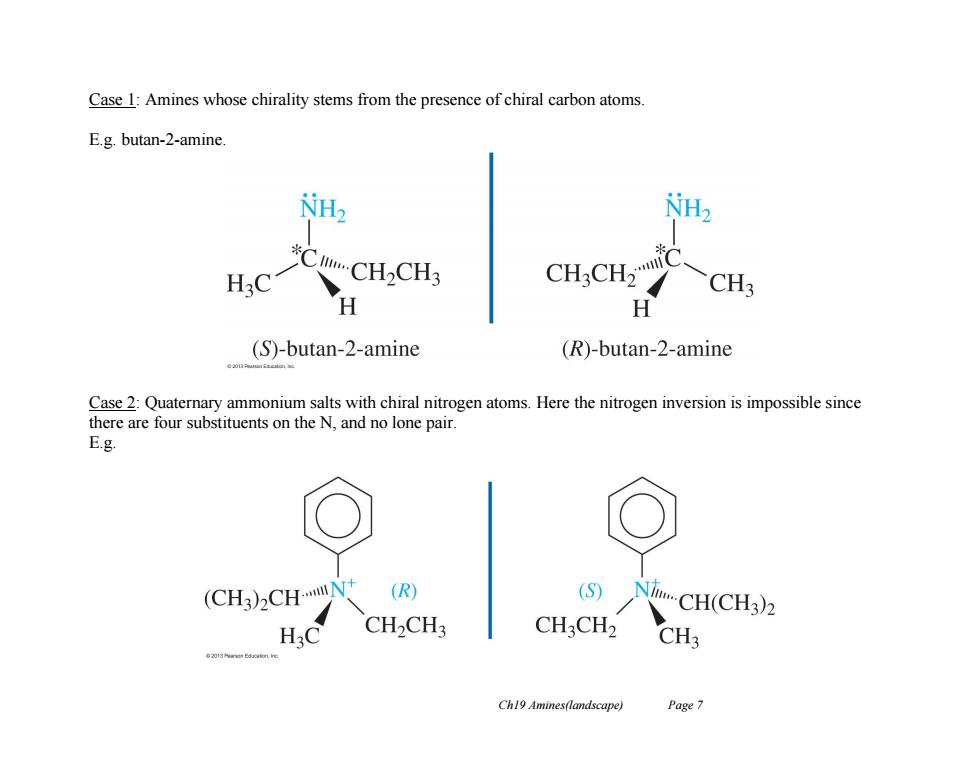
Case 1:Amines whose chirality stems from the presence of chiral carbon atoms. E.g.butan-2-amine. CH.CH CH3 H (S)-butan-2-amine (R)-butan-2-amine 020 Case 2:Quaternary ammonium salts with chiral nitrogen atoms.Here the nitrogen inversion is impossible since there are four substituents on the N,and no lone pair. E.g. (CH3)2CHN (R) (S) NiCH(CH3)2 CH2CH3 CHCH2 CH3 Ch19 Amines(landscape) Page 7Ch19 Amines(landscape) Page 7 Case 1: Amines whose chirality stems from the presence of chiral carbon atoms. E.g. butan-2-amine. Case 2: Quaternary ammonium salts with chiral nitrogen atoms. Here the nitrogen inversion is impossible since there are four substituents on the N, and no lone pair. E.g