正在加载图片...
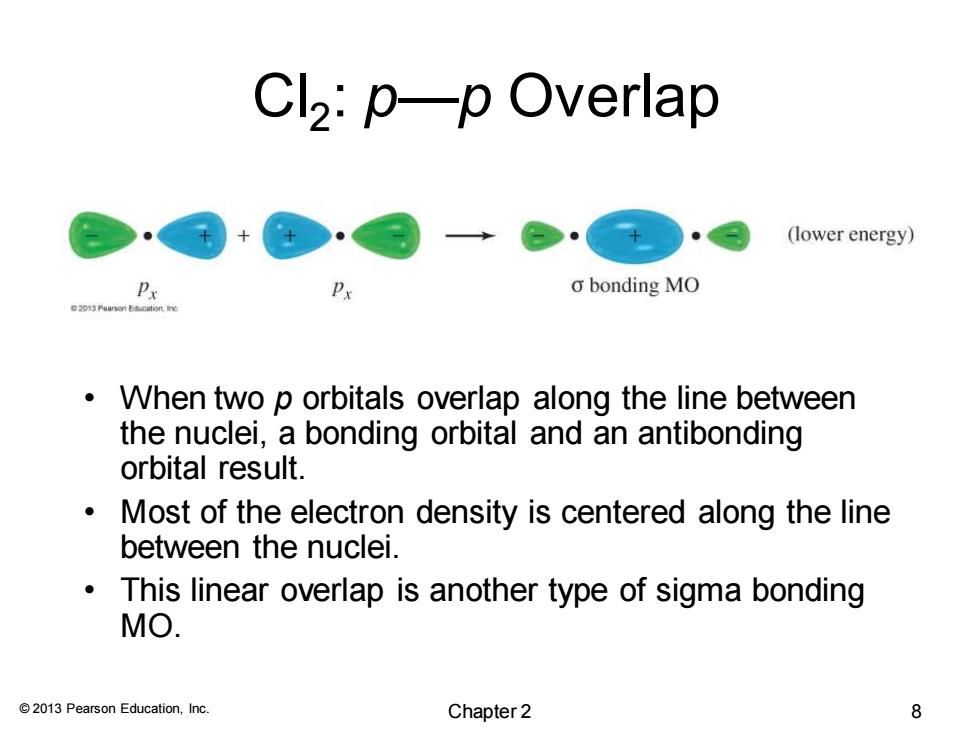
Cl2:p—p Overlap (lower energy) o bonding MO When two p orbitals overlap along the line between the nuclei,a bonding orbital and an antibonding orbital result. Most of the electron density is centered along the line between the nuclei. This linear overlap is another type of sigma bonding MO. 2013 Pearson Education,Inc. Chapter 2 8 © 2013 Pearson Education, Inc. Cl2 : p—p Overlap • When two p orbitals overlap along the line between the nuclei, a bonding orbital and an antibonding orbital result. • Most of the electron density is centered along the line between the nuclei. • This linear overlap is another type of sigma bonding MO. Chapter 2 8