正在加载图片...
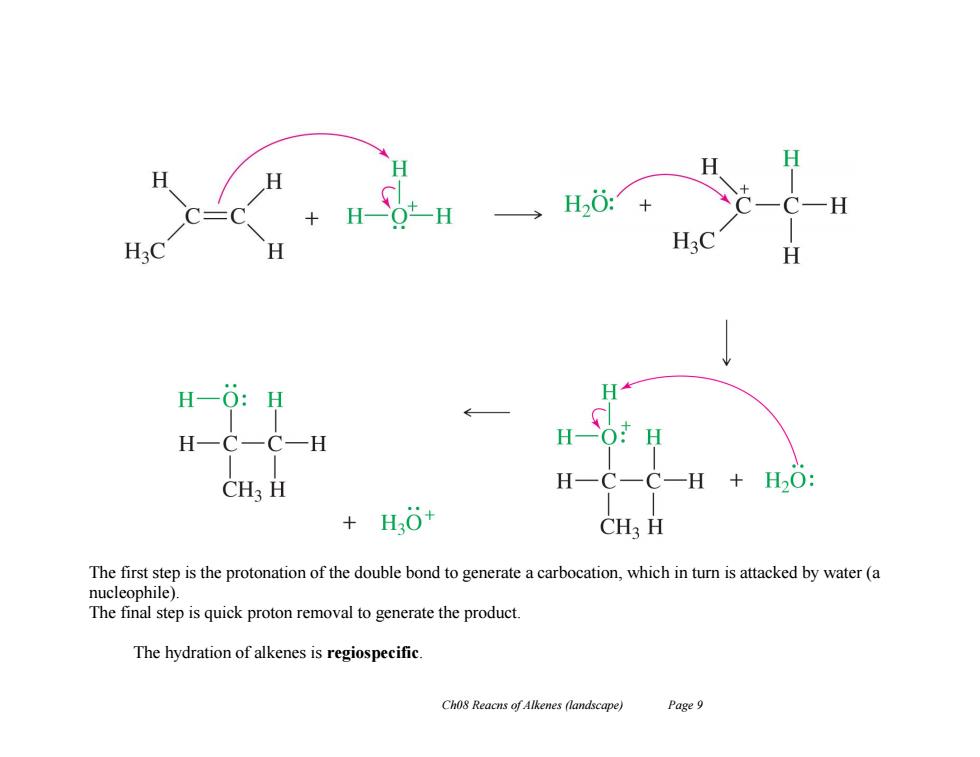
H一Ot-H H20:+ H20 H20 H H-O:H H H一C一C一H H一O:H CH3 H H-C-C一H+H2O: +H0+ CH3 H The first step is the protonation of the double bond to generate a carbocation,which in turn is attacked by water(a nucleophile). The final step is quick proton removal to generate the product. The hydration of alkenes is regiospecific. Ch08 Reacns of Alkenes (landscape) Page 9Ch08 Reacns of Alkenes (landscape) Page 9 The first step is the protonation of the double bond to generate a carbocation, which in turn is attacked by water (a nucleophile). The final step is quick proton removal to generate the product. The hydration of alkenes is regiospecific