正在加载图片...
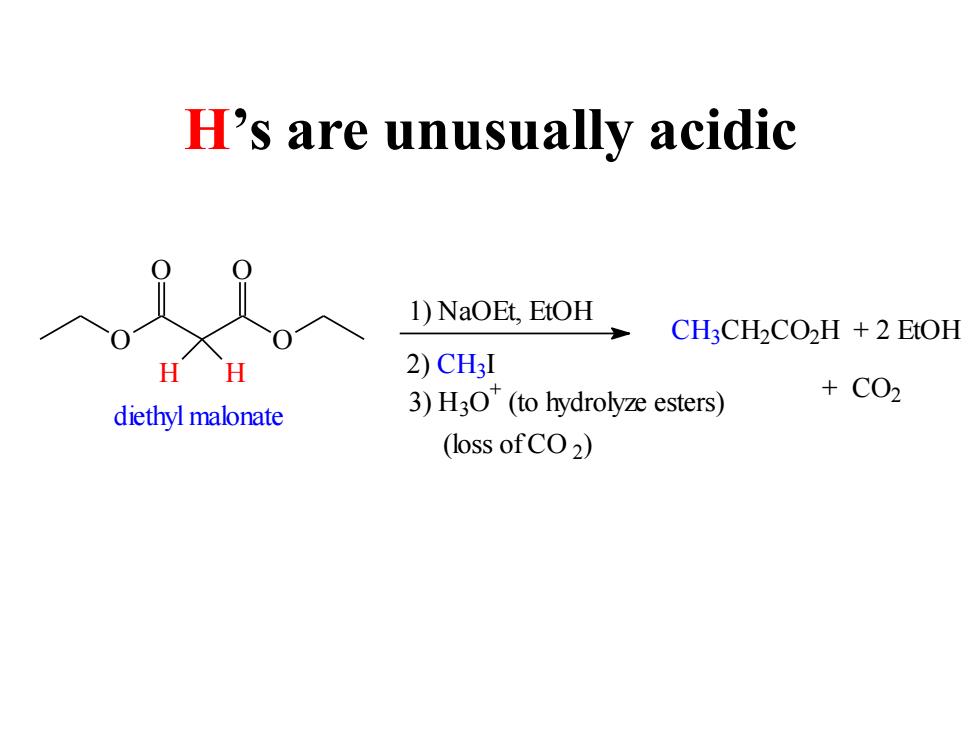
H's are unusually acidic 1)NaOEt,EtOH CH:CH2CO2H +2 EtOH H 2)CH3I +C02 diethyl malonate 3)H3O(to hydrolyze esters) (loss ofCO2) H’s are unusually acidic O O O O H H diethyl malonate 1) NaOEt, EtOH 2) C H3I 3) H3O + (to hydrolyze esters) (loss of CO 2 ) C H3C H2C O2H + 2 EtOH + CO2