正在加载图片...
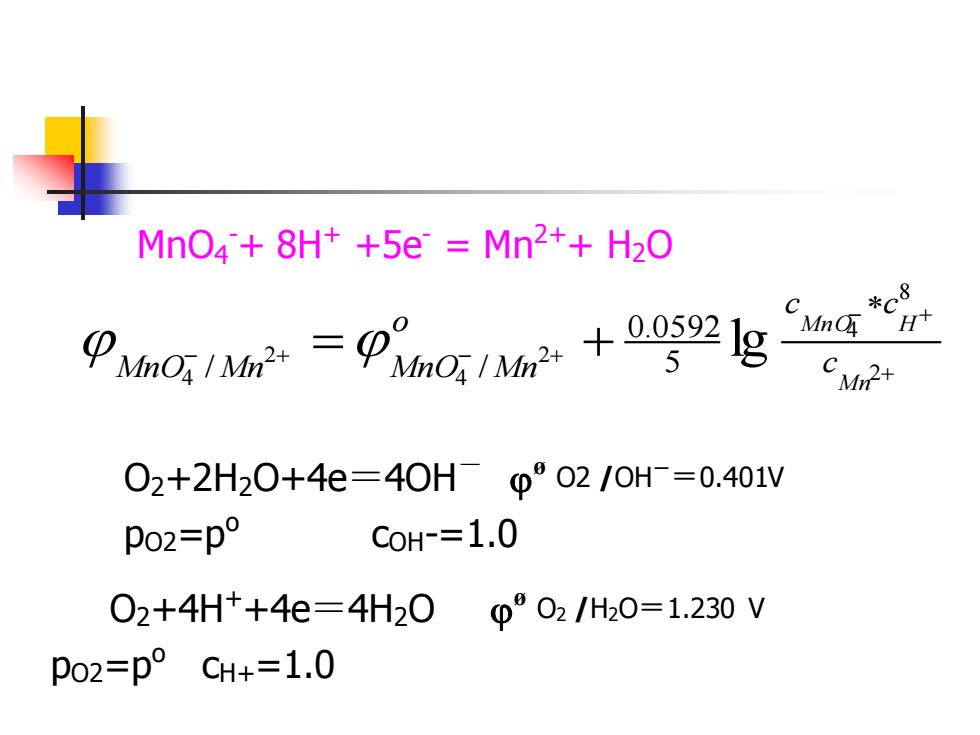
MnO4+8H++5e Mn2++H2O *c8 Pm02g Mn H C 02+2H20+4e=40Hp°02/oH=0.401V po2=p° CoH-=1.0 02+4H++4e=4H200°02/H20=1.230V po2=p°CH+=1.0 MnO4 -+ 8H+ +5e- = Mn2++ H2O O2+2H2O+4e=4OH- øO2 /OH -=0.401V pO2=po cOH-=1.0 + − + − + − + = + 2 8 4 2 4 2 4 lg 5 0.0592 / / M n MnO H c c c o MnO Mn MnO Mn O2+4H++4e=4H2O øO2 /H2O=1.230 V pO2=po cH+=1.0