正在加载图片...
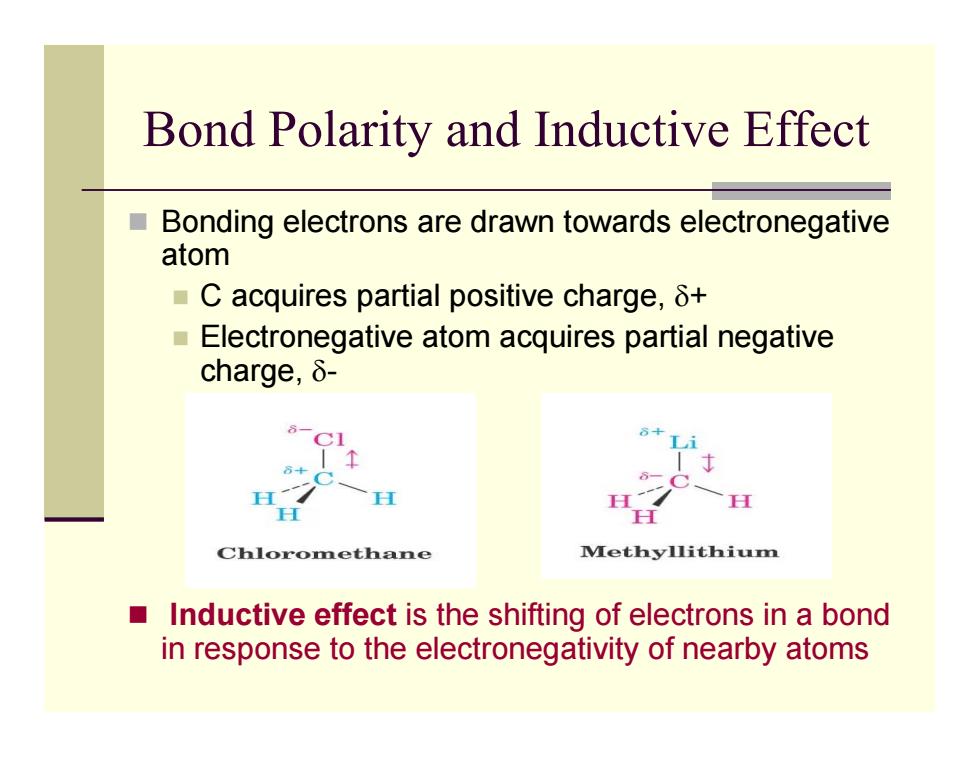
Bond Polarity and Inductive Effect ■ Bonding electrons are drawn towards electronegative atom ■C acquires partial positive charge,δt ■ Electronegative atom acquires partial negative charge,δ- H Chloromethane Methyllithium Inductive effect is the shifting of electrons in a bond in response to the electronegativity of nearby atomsBond Polarity and Inductive Effect Bonding electrons are drawn towards electronegative atom C acquires partial positive charge, δ + Electronegative atom acquires partial negative charge, δ - Inductive effect is the shifting of electrons in a bond in response to the electronegativity of nearby atoms